
Between the lines: Peter Donnelly seeks evolutionary clues hidden in our genetic code.
 The University of Chicago Magazine
Feb 1995
The University of Chicago Magazine
Feb 1995

Between the lines: Peter Donnelly seeks evolutionary clues hidden in our genetic code.
While he doesn't dig for fossils, Peter Donnelly's work may shed light on one of the more pressing issues in paleontology: How did the most recent chapter of human evolution unfold?
Rather than looking to the past for answers, Donnelly examines the DNA of living humans for traces of our early ancestors' evolution, migrations, and demographic history. It's a bit like reconstructing an epic story from the variations people make as the tale is told over and over.
Instead of the biologist's blood samples and gels striped with DNA bands, Donnelly's tools are equations, histograms, and the Sun workstation that sits beside his desk in his light-flooded Eckhart Hall office. Joining Chicago's statistics and ecology & evolution departments this past fall, Donnelly uses his mathematical training and expertise in probability theory to model the statistics of genetic inheritance-most recently, their applications to human evolution.
That modeling, he says, ranges from "fairly abstract"-studying the purely mathematical aspects of genetic change-to more specific questions, such as the likelihood of a particular evolutionary scenario. Tracing the lineage of whole populations is complex, but the basic logic isn't: If genes and other regions of DNA gather mutations at a slow, steady rate, Donnelly explains, then the extent of the differences between two individuals' DNA should provide a measure of how long ago their ancestors' family trees diverged.
Donnelly's recent work touches on a fierce debate in paleontology. Did modern humans simultaneously evolve, as one theory maintains, from interbreeding Homo erectus groups in Africa, Asia, Europe, and Australia? Or, in what has come to be known as the "African Eve" hypothesis, did a single hominid group-possibly in Africa-make that transition and then replace hominids living elsewhere? With fossil evidence inconclusive, the science of "molecular evolution" made its debut in the late 1980s, when a University of California, Berkeley-based study of mitochondrial DNA claimed that modern humans evolved on a single continent, probably Africa, only 200,000 years ago. Though flaws in the DNA evidence emerged later, the Berkeley group's "African Eve" hypothesis remains popular.
Since that study, observes Donnelly, "the fashion is to put a lot of effort on mitochondrial DNA"-found inside the energy-producing organelles of cells-because its faster mutation rate makes it well-suited to measuring recent evolution. But as an appreciation grows for what he calls the "subtleties" of molecular evolution, that view is changing-partly thanks to Donnelly's own research.
To test the "African Eve" argument and more recent mitochondrial-based claims, Donnelly-with fellow Australian Paul Marjoram of Monash University in Australia-compared geneticists' analyses of variation in mitochondrial DNA to his predictions based on a realistic mathematical model of evolution. Instead of working backward from existing data, as some geneticists have done, he began with scenarios considered plausible for life among the first Homo sapiens, including details-like the number of breeding groups and the changes in population size-that affect genetic diversity.
Projecting ahead thousands of generations, the Australians' model predicted the range of differences expected in mitochondrial DNA within small samples of people living today. Different scenarios generated different ranges, yet almost none matched the range from actual DNA samples gathered around the world by anthropologists.
Donnelly's analysis has thrown cold water on the genetic arguments both for and against an "African Eve": "My view is that there's relatively little information from just mitochondrial DNA," he says, adding that several unknowns, like the size or stability of early human breeding groups, could be affecting the observed results. "It's actually quite hard to come up with any realistic explanation for the [mitochondrial] data."
That, Donnelly says, is because the range of random mutations at one part of our chromosomes, such as mitochondrial DNA, offers just a "snapshot" of evolution-like a die tossed only once. This limitation makes it nearly impossible to see the underlying pattern left by migration and population changes-and to match that pattern to any single evolutionary model.
"Instead of looking at the mitochondria of more and more individuals," he notes, "we're much better off looking at different parts of the genome"-like the 46 nuclear chromosomes that hold most of a cell's genetic information. By studying variation within unrelated genes, "you're almost getting separate glimpses of evolution."
With Northwestern University professor Anna Di Rienzo, Donnelly hopes to do just that. They're currently analyzing more than a dozen locations on nuclear chromosomes where sequences of two, three, or four DNA bases-the letters in DNA's four-letter chemical alphabet-are known to repeat varying numbers of times. Di Rienzo, a geneticist, generated the sequences from blood samples available from tribes in Africa and South America.
Because each sequence is the same across individuals, Donnelly and Di Rienzo study mutations in the number of repetitions of those sequences-changes that pose mathematical quandaries similar to the 20- to 30-base repetitions used in DNA fingerprinting, a forensic technique that is another of his interests. For now, the two hope to use the data to unravel what biochemical mechanism-such as errors in the DNA-copying process-is behind the mutations. If they're successful, each of these DNA regions could open a new window onto human evolution.
Ahead lies the mathematical challenge of tracing lineages for what Donnelly calls the more "bizarre" characters in the human genome, like genes that jump from one chromosome to another. The science of molecular evolution still has its limits-perhaps above all, he says, the absence of a "molecular clock," or reliable measure of the rate at which mutations occur. But, he points out, models of genetic change have made huge progress in just the past five years, and may soon offer more definite answers to when, where, and how humans evolved. "The amount of data available," he notes, "is just exploding."
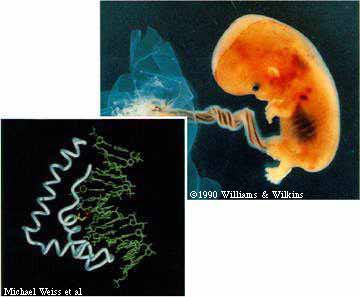
This part we already knew: Boys will be boys because they inherit a Y, rather than an X, chromosome from their fathers. But how does that lead to the more obvious, outward signs of gender? The key determinant of human maleness, it turns out, is a tiny molecular lever that reaches into a strand of DNA and pries a kink into it, setting in motion all the changes necessary to convert a sexless, 35-day-old embryo into a boy.
The discovery-reported recently in Science by researchers at the U of C, Harvard, and Massachusetts General Hospital-shows in atomic detail how SRY, the protein produced by a crucial gene on the Y chromosome, starts a molecular game of dominoes that leads to the production of testes and blocks the formation of female reproductive organs. The finding also explains how an undetected pinpoint mutation in the Y chromosome can result in the birth to parents-who, based on an amniocentesis test, had been told to expect a boy-of a healthy baby girl.
Just such a mutation was used to uncover the workings of SRY, explains Michael Weiss, a U of C professor of biochemistry and molecular biology, director of the Center for Molecular Oncology, and the study's senior author.
SRY's gene-named in 1990 for the sex-determining region of the Y chromosome-makes up less than one-half of one percent of the DNA on the Y chromosome. A mutated SRY gene produces a defective SRY protein, leading to females who are XY, or chromosomally male, and have defective gonads, but are otherwise physically normal. The condition-called pure gonadal dysgenesis-is often diagnosed when the girl fails to enter puberty, and can be successfully treated with hormones.
These women are so nearly normal, explains Weiss, because all embryos begin life more female than male. During the first 35 days of gestation, embryos of either sex develop along classic female lines and have the beginnings of a uterus, fallopian tubes, and the upper part of the vagina. Then, SRY separates boys from girls.
"If it's to be a boy, consider the challenges he faces at 35 days post-conception," says Weiss. "He has to develop male organs and get rid of the nascent female ones. The key is to make a testis"-in which one set of cells makes testosterone, which governs male sexual development, and another set produces Müllerian Inhibiting Substance (MIS), which instructs the female structures to regress.
Both processes start with the action of SRY protein. When Weiss and colleagues, including Patricia Donahoe of Massachusetts General Hospital, analyzed the protein using nuclear magnetic resonance-the same technique used by doctors to make MRI images of the body-they were able to see just how SRY does its job.
A normal SRY protein, they found, contains a long amino acid called isoleucine at a critical position, and acts by inserting the amino acid at a specific DNA sequence that helps to turn on the MIS gene. The insertion forces apart two pairs of DNA bases along the chain, bending the DNA helix, and so triggering MIS production, perhaps by altering the binding of other proteins on a nearby stretch of DNA.
But in roughly a quarter of the cases of pure gonadal dysgenesis, which occurs once in every 50,000 births, SRY's gene contains a signal error: no instruction for isoleucine. The defective SRY protein fails to grab and bend the target section of DNA, and the cell cannot produce MIS. The protein's binding fails, the researchers discovered, if the isoleucine "lever" is shortened by even a single carbon atom. It's a small change, but, says Weiss, "the effects are not subtle, and result in complete sex reversal of the child."
SRY's master switch for maleness, he notes, resembles the switches in immune cells that turn on to battle infection. "It's as if nature hit upon a solution for how to regulate a set of genes," he says, "and has used this means over and over."
Other key events in fetal development are likely to be governed by very similar mechanisms, he explains: "The process of embryogenesis is full of molecular switches as cells determine their fates. If we can understand the general switches involved in sex determination, then we can understand other molecular processes, such as how other organs develop and how cancer arises."
In the coming years, Weiss and his colleagues plan to trace factors that they believe intervene between the action of SRY and the production of MIS. Says Weiss, "We expect to find new genes that will solve the mystery of unsolved cases of human sex reversal."

In a "top ten" list of health problems among the elderly, sleep quality-according to physicians-doesn't even rank. But as Eve Van Cauter points out, the list depends on whom you ask. "If older adults are interviewed," she says, "then sleep comes as number two."
"A lot of older adults are frustrated by the fact that they can't sleep in in the morning," she explains, "and in the evening, when there are things to do, they are exhausted."
Van Cauter thinks older people are on to something. Poor sleep may not be life threatening, yet its frequent cause is, she thinks, also to blame for an array of serious health effects, including some of the physical degeneration viewed as an inevitable part of aging. A U of C research professor in the Department of Medicine who has studied connections between sleep, hormones, and biological rhythms since the late 1970s, Van Cauter thinks she's found one of the culprits-and may be fast approaching a treatment.
Not all sleep, she explains, is created equal. The sense of being physically rested comes from deep or "slow-wave" sleep, which normally fills the first hours of each night. During these hours of deep sleep, the pituitary gland secretes most of its daily output of growth hormone, produced and used by the body well past childhood.
But between ages 30 and 40, she says, both deep sleep and hormone production drop rapidly: Growth-hormone levels reach one-third to one-fourth of the amounts produced by a young adult, while slow-wave sleep-even in people who describe themselves as good sleepers-falls proportionately. "Not only is there less time spent in those deep stages," she says, "but those stages aren't as deep as they used to be."
Others have noticed the sleep-hormone link, but Van Cauter believes it's more than an association: that the drop in slow-wave sleep in fact causes the drop in hormone release. And, she applies her belief to another problem of aging. "It's thought," she says, "that the reduction in growth-hormone production is partially responsible for the changes that we have in body composition" during aging-including, for example, "the fact that, whether we stay slim or not, the ratio of fat tissue to muscle becomes higher."
Some people have wondered, she says, whether higher levels of growth-hormone production might restore strength and bone mass, making older adults "more fit, and more capable of living an independent life."
While several pharmaceutical companies are studying that prospect-through hormone injections or drugs that force the pituitary back into action-Van Cauter pursues a different approach: Why not try to restore the sleep that signals the pituitary to release growth hormone?
Although she notes that current sleep-inducing drugs make sleep more, not less, shallow, there are ways to encourage deep sleep-including exercise, as Van Cauter has demonstrated in earlier studies on how exercise and light therapy affect the body's 24-hour internal clock, which is the source of circadian rhythms and a timing system for hormone release. But while quick to recommend exercise, she feels that a new method for prompting deep sleep would still be helpful.
Recently, "totally by serendipity," Van Cauter may have found such a method. After delivering a lecture at a scientific meeting, she received a tip that a certain drug used experimentally to treat a rare disease may affect deep sleep. Her pilot study using the drug on younger subjects-in which she tried inducing deep sleep when it would not normally occur-yielded "fantastic results," she reports. "The compound restored slow-wave sleep, and it doubled or tripled, according to the dosage, the daily output of the hormone." The data, she says, show clearly that deep sleep causes growth-hormone release.
Van Cauter won't mention the drug by name-the University hopes to patent its new use-but does mention an early side effect: Her pilot-study results have helped secure long-term funding from the National Institute of Aging to study, jointly with Northwestern University, how circadian rhythms change with aging. As the project's leader, Van Cauter says the grant will put her work in an interdisciplinary context-"literally from molecular-level alterations in the central nervous system responsible for generating circadian rhythms," she explains, "all the way to alterations in sleep and wake patterns in institutionalized patients in nursing homes."
She hopes for a "cascade of health benefits" from her part in the five-year project. In addition to maintaining strength and bone mass through its role in growth-hormone release, deep sleep helps clear the body of stress hormones, produced in response to stimuli like pain, physical exhaustion, and psychological stress. And it would help older people keep their weakening internal clocks synchronized with the outside world, by strengthening the environmental cues of light-dark cycles and social schedules. Like a pacemaker for the daily rhythms of life, restored deep sleep could, says Van Cauter, "help reset the whole temporal structure of the body."
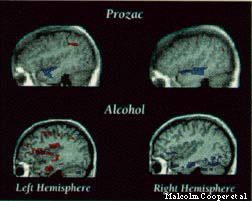
What does a drug look like in action? In studies of Prozac and alcohol, a U of C team has developed a method to see the location and extent of a drug's effect on brain behavior. The images integrate anatomical data from magnetic resonance imaging (MRI, shown in gray) with a map (in color) of drug-induced changes in glucose metabolism, a measure of cell activity produced by positron-emission tomography (PET). At left, blue regions show where the drugs decrease metabolism; red regions show areas of heightened metabolism. Radiologist and PET Center director Malcolm Cooper-who worked with Edwin Cook, Jr., of the psychiatry and pediatrics departments; Harriet De Wit of radiology; and the center's associate director, John Metz, PhD'78, of psychiatry-says the method is unlike conventional radioisotope "tracer" studies. While tracers show a drug's immediate targets within the brain, the U of C technique reveals a drug's ultimate impact: the regions responsible for its effect on behavior. Able to quantitatively measure and pinpoint that impact with image "slices" through the brain just millimeters apart, the new method should prove useful in the design of new drugs.
Why Me, Lord?

Schreiner on Job.
One of the world's oldest hard-luck stories is, says Susan Schreiner of the Divinity School, a "defining myth" of Western civilization. The biblical story of Job has fascinated writers for centuries, yet the history of their collective thought has been mostly overlooked. In Where Shall Wisdom Be Found? Calvin's Exegesis of Job from Medieval and Modern Perspectives (Chicago), Schreiner traces Job's interpreters from Gregory the Great to Kafka and H. G. Wells. "The image of Job," she says, "has challenged every attempt to explain suffering and to justify God's actions." To John Calvin-the story's most influential interpreter-Job's tale revealed an inscrutable God "whom [people] could trust despite the deepest darkness and the most awful of divine silences." The story's rich interpretive history, Schreiner says, also holds a lesson for deconstructionists: "You're not the first people that recognized that there was more than one reading to a text."
Fly By.

"All of space research up until now," says physicist John Simpson, "has been in or near the ecliptic"-the plane in which the planets orbit the sun. "Intellectually, it's like trying to describe nature while believing that the Earth is flat." That era ended this past September when the Ulysses spacecraft passed over the sun's south pole, crossing at a distance roughly twice that between the Earth and the sun. Launched in 1990, the craft first headed toward Jupiter, using the planet's huge mass to swing below the ecliptic. Now in an orbit perpendicular to the Earth's, Ulysses is headed toward the sun's north pole, which it will reach later in 1995. Among the 11 European and U.S. experiments aboard is a cosmic-ray and solar-particle investigation led by Simpson, who oversees an international team of Ulysses scientists, including Bruce McKibben of the Enrico Fermi Institute. Cosmic rays-atomic nuclei moving at nearly the speed of light-emanate from far away in our galaxy, and may help explain how stars form elements and how stars die. At the sun's poles, Ulysses can spot the rays before they are deflected by the sun's magnetic field. However, in measuring cosmic rays during the recent south-pole pass, the researchers were surprised to find that the rays' intensity was not nearly as high as expected. That, says Simpson, suggests a more complicated picture of how cosmic rays enter the solar system.
Written and compiled by Andrew Campbell.