>
> Gut
reaction: What's going on deep inside critically ill patients?
A
bacteria's drive to survive may suggest ways to help seriously
ill patients survive too.
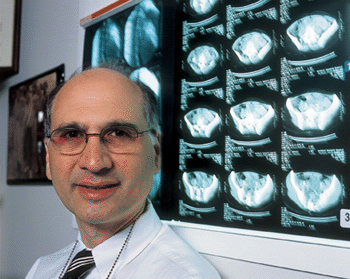
Like
many doctors, John C. Alverdy, associate professor of surgery,
recalls a critically ill patient who died in his care. In Alverdy's
case, the patient was someone he encountered during his residency
at the Michael Reese Hospital in Chicago in the mid-1980s. The
11-year-old boy, whose leukemia was in remission, had the classic
symptoms of infection: "spiking" a fever every day,
rapid heart rate, and sepsis, an inflammatory reaction to bacteria
spreading toxins throughout the body. But the microbe causing
the infection eluded doctors' efforts to identify it. Repeated
blood and urinary cultures came back negative, and CAT scans failed
to reveal a focus of infection anywhere in his body.
"What
can you do? You've taken pictures from head to toe, cultured everything
culturable, and you still can't identify the offending microbe.
Yet infection is clinically suspected, and powerful antibiotics
are usually administered," says Alverdy, now director of
the Surgical Critical Care Unit at the U of C Medical Center.
"Maybe there are unusual circumstances at work."
Among
intensive-care patients in whom infection is suspected on clinical
grounds, as many as 50 percent have no identifiable source. Alverdy
has set about reconstructing the circumstances that cause sepsis,
currently blamed for the deaths of 1,400 patients each day worldwide,
making it the No. 1 killer in intensive-care units (ICUs). A wiry
guy with a shiny pate and rectangular eyeglasses, the surgeon
focuses on the one place in the body that's full of bacteria-the
gut. With a $1.1 million grant from the National Institutes of
Health, Alverdy is showing that Pseudomonas aeruginosa,
a bacterium commonly found in the intestinal tract of critically
ill humans, can mount the equivalent of "guerrilla warfare"
if its survival is threatened. And he believes he's found a way
to inhibit-or at least make peace with-the bacteria in what he
calls a classic evolutionary struggle to survive.
Why
would a bacterium that can naturally live in our intestinal tracts
want to kill us? Because artificial support systems in the ICU
create what Alverdy calls a "historically unprecedented"
situation, forcing a bacterium to resort to unique tactics to
gain nutrition and survive.
"Bacteria
may use systems analogous to smell, taste, and feel to assess
the body's response to injury and to gauge whether survival is
likely." For example, Pseudomonas has virulence genes
activated by high levels of the stress hormone norepinephrine,
which the body produces after major surgery. The norepinephrine
tells the bacterium that the host is under extreme duress, which
may mean death is imminent for colonizing bacteria too. But since
a dead host also may mean immediate access to months and years
of food in the form of decaying tissue, "it's in their best
interest to kill you," to temporarily stave off their own
demise.
The
ICU isn't helping the situation by feeding patients "chemically
defined foodstuff" intravenously or directly into the intestines.
When humans are healthy, says Alverdy, they treat their intestinal
microbes to a predictable environment and a constant food supply,
sending the by-products of digestion and vital nutrients down
to their intestines for the microbes to eat. But IVs and chemically
defined food deprive the bacteria of their customary nutrients.
After a long stay in the ICU, the bacteria in a patient's gut
get hungry.
Meanwhile,
another "unusual" circumstance may be prompting bacteria
to increase their potential for harm. It's possible, says Alverdy,
that the ICU environment itself inadvertently creates a situation
in which powerful bacteria such as Pseudomonas flourish.
Large doses of antibiotics, steroids, and antacids upset the balance
of microbial "flora" in patients' digestive tracts,
he says, "wiping out the good organisms and letting pathogens
like Pseudomonas multiply."
Alverdy
cites recent research demonstrating that Pseudomonas, like
other bacteria, efficiently monitors its environment, communicating
along a "quorum-sensing signalling system"-a cell-to-cell
game of postman in which the chemical homoserine lactone gets
produced and absorbed down a line of organisms, enabling the bacteria
to track population density. A "critical mass"-about
105 organisms, speculates Alverdy, perhaps hiding out in the pouch-like
cecum of the large intestine-is necessary before the bacteria
mount a toxic offensive. Alverdy's lab has shown that if Pseudomonas
achieves a critical mass while on red alert from norepinephrine,
it expresses the gene PA-I, which then allows the organism to
penetrate the intestinal walls with adhesins-little "molecular
syringes" that deliver shots of toxins into the host's cells.
On
the light box in Alverdy's office hangs a series of transparent
"blots" showing Pseudomonas before and during
PA-I expression. The blots look like inked fingerprints-some light,
others dark and heavy. In fact, the blots come from the stool
of mice in which Alverdy simulated the hostile environment of
a critically ill patient's gut. He performed major surgery on
the mice, removing 30 percent of their livers. After putting the
animals on a 48-hour water-only diet to simulate a postoperative
state, he injected a critical mass of Pseudomonas into
the animals' ceca and regularly swabbed their intestinal linings.
As the bacteria's quorum-sensing system began producing and absorbing
homoserine lactone molecules and the PA-I genes were expressed,
the blots got progressively darker. In 90 percent of the mice,
the PA-I attack was lethal.
Alverdy
describes such gene expression as guerrilla warfare because it
doesn't follow conventional thinking on how bacteria kill their
hosts. "Ask any infectious-disease specialist, and they'll
tell you that most bacteria kill their host by disseminating through
the body, eventually causing bloodstream infection. An area of
the body gets infected, it gets inflamed and full of pus, the
bacteria and its toxins become bloodborne, and the patient dies
because their immune system is overwhelmed." But Alverdy
argues that bacteria whose survival is threatened have nothing
to gain by going up against the immune system. Instead, he believes
they use extension appendages such as adhesins to feed directly
off the intestinal host cells. "If this resource is threatened,
they are capable of delivering toxic shots en masse to undo the
host.
"The
most amazing thing is that this can occur without clinical detection.
The bacteria use subterfuge to kill, without necessarily invading
the bloodstream." It isn't yet clear how, but Pseudomonas
is able to touch the intestinal wall and stimulate the opening
of natural "garage doors"-gaps in the intestinal lining
that let nutrients in and, in theory, keep toxins out. The doors
open, and the appendages shoot their toxins into the cells. Evidence
suggests that bacteria can accomplish the sneak attack by secreting
immune-suppressing substances to camouflage themselves from host
recognition.
Alverdy
proposes two ways for ICU physicians to prevent a potentially
lethal attack in the gut. Routinely blotting the intestinal linings
of critically ill patients would tell physicians when bacteria
are reaching critical mass. With the NIH grant, Alverdy is studying
a natural sugar that could be used to blanket bacteria and prevent
them from poking cells with their toxic needles.
A
second way to fight the microbial guerrillas is simple: insert
food into the bowels of critically ill patients in a form that
the microbes can eat. Says Alverdy, "All these bacteria want
is a normal environment and a healthy host." It's a small
price for peace.-S.A.S.

![]()
